What is a Corrective Action?
The corrective action process aims to establish the process for identifying, documenting, and analyzing nonconformities and mitigating their impacts by implementing appropriate actions.
An organization’s quality management system must be geared toward proactively eliminating actual and potential deficiencies. Repeated nonconformities in processes, products, and services must be investigated, and action must be implemented to prevent their occurrence.
Contents
- What is a Corrective Action?
- Corrective Action Vs Preventive Action?
- What Does ISO 9001 Require For Nonconformaties?
- What Are The 6 Steps for Successful Corrective Action?
- What Is A Corrective Action Report?
- What Is A Corrective Action Tracker?
- Corrective Action Plan
- What Is A Corrective Action Process Map?
- Summary
- Corrective Action Template
Don’t Try to Manage It All Alone!
Corrective Action Vs Preventive Action?
A Corrective Action is re-active — deals with the problem AFTER the event. Corrects it.
A Preventive Action is pro-active — deals with the problem BEFORE it happens. Prevents it.

What Does ISO 9001 Require For Nonconformaties?
ISO 9001 Clause 10.2 nonconformity and corrective action requires businesses to react to nonconformities by controlling and correcting the problem. The clause states the requirements for when a nonconformity occurs and includes requirements for actions to prevent similar problems from occurring.
Organizations must take whatever action is necessary to control and correct the nonconformity and to deal with any resulting impact by determining what caused the nonconformity and considering whether the potential for a similar problem remains.
This is done by considering whether any further corrective action is required to prevent a similar nonconformity from arising at the same place or somewhere else in the future, and determining if similar nonconformities have occurred elsewhere and whether similar corrective action needs to be taken.
Taking appropriate action to address the effects of the problem may require a simple correction by the process owner or operator where it was discovered, or if a major failure or defect exists, more significant levels of resources would be needed for problem-solving and corrective action.
Any nonconformities and their subsequent actions to prevent reoccurrence, as well as the actions to monitor the effectiveness of the corrective action(s), should be documented and retained. Therefore, consideration should be given to developing and using the Corrective Action Tracker to capture this information.
What Are The 6 Steps for Successful Corrective Action?
This article presents a reliable 6-step methodology for meeting the clause's requirements. The following steps will help you create a successful corrective action process:
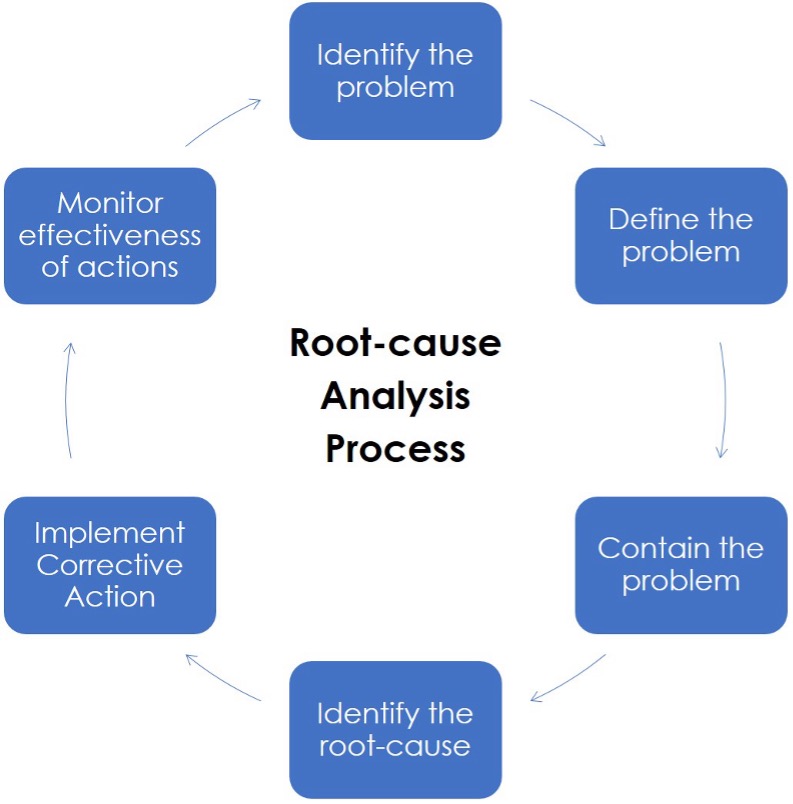
Step 1: Identify the Problem
The following guidelines are for completing root cause analysis and corrective action responses to audit nonconformity reports (NCRs). However, these guidelines should also be applied to corrective action responses from other sources (customer complaints, internal defects, etc.)
Once a problem has been identified through inspection, customer complaints, or audit results, it should be captured using nonconformity reports (NCRs) or corrective action reports (CARs) to identify who is affected by the problem and what the impact is.
Considering the following:
- What are the operations, products, materials, defects, and malfunctions that may characterize the problem? What is it about?
- Who is concerned with the problem? Who is reporting the problem? Who is rectifying the problem? Who is the problem affecting?
- Where is the event's location: shop floor, services, machine, process step?
- Where is it seen? Where does it originate?
- When does the event appear (time, date, when does it start, how long does it last, how often)
- When is the problem reported as defective? When is the problem repaired?
- Has it occurred before? If yes, what is the history?
- How do we know there is a problem (how is it detected)?
- How does the event appear, and how does it stop?
- How frequently is the problem experienced?
- How is the effect of the problem being measured (costs, delays, scrap rate, complaints, etc)?
- How is the problem currently addressed? How is it corrected?
This step helps to fully describe a situation, precisely analyze all its elements, and gain a shared understanding of them, allowing the definition of an action plan. Ensure that all team members agree on the definition of the issue and its resulting impact.
Appropriate statistical and non-statistical techniques can be applied to analyze the nonconformity. The organization will select the appropriate methodology based on the problem's complexity.
Examples of statistical techniques are:
- Statistical Process Control (SPC) charts
- Pareto analysis and data trending
- Linear and non-linear regression analysis
- Experimental design (DOE – Design of Experiments) and analysis of variance
- Graphical methods (histograms, scatter plots, etc.)
Non-statistical techniques include:
- Management reviews and the results from meetings
- 5W2H: Who, what, where, when, why, how, and how many for both occurrence and non-detection
- Product review committees (internal/external)
- Failure Mode and Effect Analysis (FMEA)
- Fault Tree Analysis (FTA)
The Quality Manager will often detect and determine the extent of the problem (place, time, quantity, number of affected components, versions, projects, vehicles, Customer sites, etc.) to check whether the problem is relevant to safety and if relevant stakeholders need to be informed.
Start with Expert Templates, then Make Them Yours
Step 2: Define The Problem
Describe the problem by identifying what is wrong and detailing the problem in quantifiable terms. Get a clear picture of the problem (based on the available data from internal or external sources, including photos and videos). Define the goals that need to be achieved (as a resolution).
The problem description should describe the problems in terms of what, where, when, and how big. On a flip chart, presentation board, or even paper, describe what you know about the problem. Try to document the problem and describe it as completely as possible.
The description should contain facts, such as observations and documentary evidence, not assumptions. All information must be gathered before identifying the root cause can begin. Ensure the above factors are true before moving to the next step.
Consider any new information the team may have gathered since completing the initial problem description. Define, verify, and implement the interim containment action to isolate the effects of the problem from any internal/external customer until Permanent Corrective Actions (PCA) are implemented.
Now that you have narrowed down the possible root causes, you need to develop theories about how the problem occurred. Theories are statements that describe how a change may have created the problem. To develop root cause theories, brainstorming techniques are used to generate ideas.
Establish A Response Team
Identify representatives from functions that may have an influence on the corrective action process, including the identification of the root causes. Remember to assign responsibilities and objectives to the team members.
Remember, those performing the job, such as operators, inspectors, drivers, etc., are the best people to help identify the actual causes; do not leave them out of the team!
The size and composition of the team should depend on the complexity and the impact of the problem. The team's composition is not fixed forever and may evolve depending on the analysis results and the required actions. New team members should join the team if the analysis shows they are identified as being in the scope; others will leave if their area is out of the scope.
However, consideration should be made that expanding the size of the core team to over 6 to 8 members generally results in less efficiency. When more members or special skills are required, sub-teams should be considered. Do not forget that root cause analysis must not be used to assign blame or transfer responsibility. In summary, you should establish an investigation team with:
- Process and/or product knowledge
- Allocated time and resources
- Authority to solve the problem and implement corrective actions
- Skill in the required technical disciplines
- A designated Team Leader
Brainstorming sessions should be used to identify potential causes and investigate each potential cause. Coordinate parallel activities with different team members to help expedite the verification process. Top management should ensure that the team has the appropriate resources (e.g., time, skills, tools) to handle the described problem.
Make sure that Top management is informed and involved in resolving the problems. The organization should define an escalation process based on problem criticality (From safety-related to a single-case problem not affecting operation).
Undertake A Comparative Analysis
Once you have reviewed the problem description, you can undertake a comparative analysis. A comparative analysis will help you identify relevant changes in a change-induced situation. Then, you can reduce the number of possibilities that you must consider to determine the root cause.
To complete a comparative analysis:
- Ask yourself: what is unique, peculiar, different, or unusual about the symptoms?
- Consider features such as people, processes, materials, machines, and the environment
- List all facts without prejudice as to the possible cause
Consider each difference you listed, and look for changes; ask yourself:
- What has changed to give rise to this difference?
- Keep in mind that each difference may not have a corresponding change
- List the changes next to the difference
- Look at the dates each change occurred
- Eliminate some changes if they happened after the problem started
- Consider categories of people, machines, processes, or measurements
If the problem is change-induced, the root cause must result from a change relative to one or more identified changes. It is important to remember that you have not yet moved from the ‘observations’ phase of the process.
Any information you develop during the comparative analysis must be fact-based, not opinion-based, and valid only for the symptom’s information. Do not rule out any facts that might be valid answers. Write it down if it is a fact and it answers the question.
The problem description should describe the problems in terms of what, where, when, and how big. The description should contain facts; essential information such as observations and documentary documentation as evidence, and not assumptions. All information must be gathered before the formal process of identifying the cause can begin.
Ensure the above factors are true before moving to the next step. Consider any new information the other team member may have gathered since completing the initial problem description.
Communicate The Problem
Continual communication between all team members is mandatory, for instance, through regular reviews until all members identify and agree upon the root cause. Inside the organization where the problem originated, all supply chain actors are to ensure practical root cause analysis and definitive corrective action implementation.
A team leader needs to know that he has been chosen and why he has been assigned this role, along with a description of the team’s objectives and constraints, while each team member needs to understand his role and objectives.
The line managers of each team member need to know each member’s level of involvement, e.g., their time, duration, and role. All stakeholders must be informed of the team’s composition and objectives. Root causes must be communicated to all stakeholders and agreed upon, especially by the customer when he is impacted.
Communication needs to occur internally, between various tier levels, and between the supplier and the customer to immediately stop the problem from worsening, ensure a complete understanding, and verify that implemented solutions are satisfactory.
Jump Start Your Documentation
Step 3: Contain The Problem
Select An Interim Containment Action
Your organization should first contain the problem by taking immediate corrective action (ICA) and then evaluating the need to initiate the formal problem-solving process. Where necessary, provide an emergency response action to protect the customer from the problem, protect the customer operations and the organization (to stop the problem from worsening), and verify that the problem does not degrade until the root causes are known.
Take immediate corrective action to eliminate, prevent, or reduce the probability of any additional non-conformances related to the apparent cause from happening again in the short term while the investigation is ongoing by:
- Identifying, isolating, and performing immediate correction of all defective parts or data
- Identifying apparent cause and performing immediate corrective action to eliminate, prevent, or reduce the probability of any additional non-conformances from happening again in the short term
- Typical immediate containment actions may include:
- Immediately stopping the working process
- Stopping all deliveries
- Recalling the affected product (still within the organization or that has already been delivered)
- Over-inspecting increases the inspection burden and adds additional criteria
- Identifying immediate, potential risks for the same parts/batch if not detected
- Are you able to assess criticality when determining apparent criticality? If not, who should inform the customer, the design office, and certificate holders to assist in evaluating the criticality?
During these sorting operations, the organization should pay particular attention to the traceability of the bad and good parts. This also offers the chance to identify and inform the affected customers.
The organization should define a transparent identification system for the parts sorted and inspected (e.g., green label, specific marking) and prepare a summary table giving an exhaustive overview of the results regarding the quantity of good and bad parts per production date.
Matching this inspection information with the production information (assembly traceability, e.g., equipment used, specific teams/shift, provider source) should provide valuable information to confirm or inform the hypothesis during root cause analysis.
Note: These actions may be temporary and should remain in place until root cause(s) are identified, and permanent root cause corrective actions are implemented and verified to be effective. Immediate containment actions terminate when corrective actions are in place or when the study has found a more effective containment action. Immediate containment actions must have an agreed-upon, effective life span.
Verify the Interim Containment Action
An interim containment action can be any action that protects the customer from the problem. However, before you implement an interim containment action, you need to verify that the interim containment action will work.
To verify the interim containment action:
- Prove before implementation that it protects the customer from the problem
- Provide a before-and-after comparison
- Prove that the interim containment action will not introduce any new problems
Methods of verification may include:
- A test to determine the desired performance level
- A demonstration that changes eliminated the issue without creating a new problem
- A comparison between the interim containment action and similar proven actions
- A review to evaluate whether the interim containment action was effective
- Ensure that the interim containment action did not introduce a new problem
Any interim containment action you implement must protect the customer from the problem without introducing any new problems. Also, a single interim containment action may not be enough. You may need to implement more than one interim containment action to protect the customer entirely.
Why Reinvent The Wheel?
Step 4: Identify The Root-cause
Root cause analysis (RCA) is a problem-solving method that identifies the root causes of problems or events.
The practice of RCA is predicated on the belief that the problems are best solved by attempting to correct or eliminate root causes instead of merely addressing the immediately obvious symptom.
All problems have at least two root cause categories, the cause of occurrence. The reasons for the problem and the cause of non-detection, e.g., the problem not being detected while it was generated.
The root cause analysis is a questioning technique that goes to where the problem started and focuses on facts and data. It is a technique that explores the hidden causes of the specific problem using descriptive techniques (e.g., A3, 5Y, and 8D for both occurrence and non-detection).
Working with assumptions during root cause analysis might be necessary first, but these assumptions must be proven. Several techniques are available to determine potential causes (e.g., 5-Why, Fishbone Analysis (Ishikawa), Factor Tree Analysis).
Isolate and verify the root cause by testing each possible cause against the problem description and test data, then isolate and verify the place in the process where the effect of the root cause should have been detected and contained: the escape point.
Directing corrective measures at root causes will minimize the likelihood of the problem’s recurrence. The general principles of root cause analysis include:
- Aiming performance improvement measures at root causes is more effective than merely treating the symptoms of a problem;
- To be effective, RCA must be performed systematically, with conclusions and causes backed up by documented evidence;
- There is usually more than one root cause for any given problem;
- To be effective, the analysis must establish all known causal relationships between the root cause(s) and the defined problem;
- General process for performing and documenting an RCA-based corrective action.
Note that root cause analysis forms the most critical part of successful corrective action because it directs the corrective action at the root of the problem. That is to say, it is effective solutions we seek, not root causes.
Root causes are secondary to the goal of prevention and are only revealed after deciding which solutions to implement.
Using The A3 Action Plan
The A3-Action Plan is most effective in addressing small to medium-sized problems and structuring improvement projects. Aim to complete the A3 action plan within 15 days.
When the issue is minor and the solution is obvious, a formal analysis is not required; the A3 method defines what to do, who to do it, and when. This is intended to be a ‘quick fix’ to be applied within one day to low-risk issues, contain and correct snags and minor issues, and structure improvement projects.
- A problem-solving approach–built around PDCA
- A concise summary of the problem and solution
- A way of structuring thinking
- A communication tool for workers to report problems and improvement suggestions to management
- A way for management to structure and provide discipline to the improvement process
- Used for any kind of problem in all parts of the business
Briefly state the problem in one or two sentences. The problem statement includes what is being affected and where it is occurring, including background and current conditions:
- What is the business reason for choosing this issue?
- What is the problem, need, or gap in performance?
- How often does it happen?
- Is there a pattern of occurrence? Quantify the extent and causals
- What specific conditions indicate a problem or need, where, and how much?
- Show the facts visually with run charts, graphs, and maps
- Ensure the problem statement is specific
- Ensure the problem statement does not include an implied solution
- Ensure the problem statement states “what,” not “why.”
- Ensure the problem statement does not include goals
What goals would you like to see based on resolving some of the contributing factors of the problem? How much improvement? By when? Metrics?
Using The 5-Whys Analysis Process
Use 5-Whys for troubleshooting, quality improvement, and problem-solving, but it is most effective when used to resolve simple or moderately complex problems. Aim to complete the 5-Whys analysis within 30 days.
Invented in the 1930s by Toyota Founder Kiichiro Toyoda’s father, Sakichi, and made popular in the 1970s by the Toyota Production System, the 5-Whys strategy involves looking at any problem and asking, ‘Why?’ and ‘What caused this problem?’
The 5-Whys technique offers some real benefits to organizations with varying degrees of management system maturity:
- Simplicity. It is easy to use and requires no advanced mathematics or tools, which allows you to dig deep and find underlying issues rather than using quick-fix solutions
- Effectiveness. It helps to separate the symptoms from the causes and identify the root cause of a problem using evidence-based analysis
- Comprehensiveness. It aids in determining the relationships between various problem causes and allows you to eliminate problems for good proactively
- Flexibility. It works well alone and when combined with other quality improvement and troubleshooting techniques such as the ones listed above
- Engaging. By building a culture that embraces progress, by its very nature, it fosters and produces teamwork within and outside of the organization, and encourages the reporting of issues without fear or judgment
- Inexpensive. It is a guided, team-focused exercise that seeks to improve and adapt processes to ensure long-term success. There are no additional costs
The idea is simple: by asking the question ‘Why,’ you can separate the symptoms from the causes of a problem. This is critical as symptoms often mask the causes of problems. As with effective incident classification, basing actions on symptoms is the worst possible practice.
Essential Elements Of The 5-Whys
When performing root cause analysis, always address why the nonconforming product was produced and why it was passed on to the internal or external customer; in other words, ‘why was it made and why was it shipped?’. Using the technique effectively will define any nonconformances' root causes and lead you to define practical, long-term corrective actions.
- The question “Why did this problem occur?” is a single-layer question, like peeling an onion
- The strategy of questioning must be aligned with the nature of cause-and-effect systems
- Questions must be carefully structured to yield conclusive answers
- Each layer must involve a carefully constructed split of the system such that the approach converges on the causal mechanism
- Sound experimental approaches are required to answer the question correctly
- Asking “why” 5 times is a rule of thumb, not a precise requirement
The answer to the first ‘why’ often uncovers another reason and generates another ‘why.’ It often takes five ‘whys’ to arrive at the root cause of the problem. You will probably ask more or fewer than 5 ‘whys’ in practice. Remember the following key phrase as a background thought during any 5-why exercise: ‘People do not fail; processes do!’.
How To Use The 5-Whys
The 5-Whys strategy is a simple, effective tool for uncovering the hidden causes of a specific problem and is used in troubleshooting, problem-solving, and quality-improvement initiatives. Brainstorming sessions should undertake the 5-Why for the cause of the problem and develop a separate 5-Why for how the issue escaped local control points to the customer.
Brainstorming rules are as follows:
- Focus on the problem by repeating the problem statement often
- Listen carefully for ‘evaluative comments’ and stop them immediately
- Reward all team members for their input
- Clarify potential causes if they are unclear, but don’t do the clarifying for someone
- If someone is having difficulty, review the ideas already mentioned
- If they are still having difficulty, move on to avoid frustrating them
Note: When brainstorming, the quantity of ideas is more important than the quality of the ideas generated.
- Assemble a team of people knowledgeable about the area of nonconformance. Include as many personnel as possible
- On a flip chart, presentation board, or even paper, write out a description of what you know about the problem
- Try to document the problem and describe it as completely as possible. Refine the definition with the team and agree on the definition of the problem
- Have the team members ask ‘why’ the problem as described could occur, and write the answer down underneath the problem description
- If the answer provided from 3 (above) does not solve the problem, you must repeat steps 3 and 4 until you do
- If the answer from 3 (above) seems likely to solve the problem, ensure the team agrees and attempts a resolution using the answer. You may find that there is more than one root cause of the problem
- The root cause is the lowest level of cause that can be counter-measured
The 5-Whys can help you uncover root causes quickly. However, making a single mistake in any question or answer can produce false or misleading results.
If you discover more than one root cause for each nonconformance, corrective actions should also be implemented for each of these.
Mastering The 5-Whys
To validate those potential root causes under your control, you can apply the following validations to your answers or root causes. Ask the following questions for every possible root cause you identify at all levels of the 5-Whys:
- Is there any proof, something you can measure or observe, to support the root cause determination?
- Does any history or knowledge indicate that the possible root cause could produce such a problem?
- Is there anything “underneath” the possible root cause that could be more probable?
- Is there anything this possible root cause requires to produce the problem?
- Are there any other causes that could produce the same problem?
Example 1 - Nonconformance: Components are being delivered late to our customers.
- Why 1 - Why were we unable to meet the agreed-upon timeline or schedule for delivery? The job took much longer than we thought it would
- Why 2 - Why did it take so much longer? Because we underestimated the complexity of the job
- Why 3 - Why did we underestimate the complexity of the job? Because we made a quick estimate of the time needed to complete it, and did not list the individual stages required to finish the project
- Why 4 - Why didn’t we do this? Because we were running behind on other projects
- Why 5 - Why are we running behind on other projects? Root cause - We do not allow enough manufacturing/lead time when issuing quotations to your clients
Example 2 - Nonconformance: The machine keeps failing.
- Why 1 - Why did the equipment fail? Because the circuit board burnt out
- Why 2 - Why did the circuit board burn out? Because it overheated
- Why 3 - Why did it overheat? Because it wasn’t getting enough air
- Why 4 - Why was it not getting enough air? Because the filter wasn’t changed
- Why 5 - Why was the filter not changed? Root cause - There was no preventive maintenance schedule informing the operator to do so
In this example, the fifth why above suggests a broken process or behavior that can be altered and measured for effectiveness
| 5-Whys analysis to determine the root cause of the problem: |
| Identify the root cause using the 5-whys technique. Root cause analysis is mandatory for corrective action, but is optional for OFIs and suggestions. Examples of possible contributing factors to be considered when doing the 5-whys root cause analysis are shown. |
|
Why did the problem occur? What is the root cause of the technical problem that caused the nonconformance? (Examples: Inadequate tooling, fixturing, gauging, handling, and work instructions.) |
| 1st. Why: |
| 2nd. Why: |
| 3rd. Why: |
| 4th. Why: |
| 5th. Why, and Root cause: |
|
Why was the problem not detected? How did the nonconformance escape the process? Why did the quality system fail to detect the nonconformance? (Examples: inadequate in-process/final inspection, testing, process control) |
| 1st. Why: |
| 2nd. Why: |
| 3rd. Why: |
| 4th. Why: |
| 5th. Why, and Root cause: |
Root causes are physical causes and are uniquely related to the specific object experiencing the problem. They are likely related to one or more of the causes listed below. The causes and effects can be mapped using a fishbone analysis diagram to gain a shared insight into the problem, develop possible solutions, and create a snapshot of the team’s collective knowledge.
Search for possible actions to eliminate the root cause and thus solve the problem. All defined corrective actions need to be linked to the root causes. List the corrective actions and select them according to the time to resolve them and the implementation efficiency.
Validate The Findings
Your organization should validate the answers to root causes by testing the logic of the 5-Whys from the first one to the last one, and then using a reverse approach to validate the logic using the ‘Therefore’ from the last Why to the first one.
Using The 8D Analysis Process
The eight disciplines (8D) are commonly used in automotive, and the problem-solving process (PSP) in avionics. Both methods are similar, using eight steps to address complex problems, focusing on a fast reaction to nonconformances, and completing the first three steps within three days. Aim to complete the 8D exercise within 60 days.
D0: Prepare For Problem-solving
Awareness of a problem symptom may come from feedback from the field, repair centers, through failure analysis, or negative trends in metrics (e.g., PPM). An 8D is typically warranted when the cause of the problem is unknown, the complexity exceeds the ability of one person, and the priority is significant enough to fix the problem at the root cause level and to prevent recurrence.
Initiate the 8D process when a responsible function becomes aware of a problem symptom that warrants an 8D. Define and quantify the problem symptom as experienced by the customer, using the customer's words and terminology. Identify the customer (s) and other parties affected by the issue.
Determine if Emergency Response Action (ERA) is required. ERA is directed toward a product already in the field and is intended to protect the customer from any additional impact from the problem symptom.
If an ERA is necessary, select an appropriate action, develop an implementation plan, implement it, and validate its effectiveness. Key D0 Elements
- Define and quantify the symptom
- Identify the customer and affected parties (e.g., distributors, etc.)
- Emergency Response Action (if necessary)
D1: Establish A Team
Appoint a team champion with the authority to make process changes, make resources available, and monitor team progress. Appoint a team leader who will direct the team, use the 8D methodology, and coordinate team activities with the business and its functions.
Select cross-functional team members based on their skills, technical knowledge relative to the problem, and ability to represent the stakeholders involved.
Key D1 Elements:
- Identify a champion
- Identify a team leader
- Select team members
D2: Problem Description
Create a problem statement that answers “What is wrong with What?” (e.g., Product XYZ fails in a brittle fracture mode under installation torque).
Perform a failure analysis to understand intermediate causes and provide details where feasible. Include pictures, sketches, or marked-up drawings of the issue.
Add detail through an Is/Is Not Analysis, answering the questions What?, Where?, When?, and How Big? Understand the process through a process map documenting the operations and control points of the process.
Additional tools that may be useful are a timeline, cause/effect diagram, drill-down, concentration diagram, and statistical analyses.
Create a formal problem description by adding additional information to the problem statement. The problem description defines the boundaries of the problem in terms of what it is and what is not, but could be.
Key D2 Elements:
- Problem statement
- Failure analysis
- Is/Is Not analysis
- Process flow with control points identified
- Problem Description
D3: Interim Containment Actions
The purpose of Interim Containment Actions (ICA) is to protect the customer from further effects of the issue, to limit exposure and potential liability. ICAs may include suspension of the process and/or shipments, 100% inspection, sorting, rework, substitution, or additional operations.
Consider stock at or in transit to customer locations, finished goods warehouses, subcontracted service providers, and in quarantine.
Notify personnel in the area from which the nonconformance originated that the nonconformance has occurred, including written instructions and visual aids as appropriate.
Choose the best ICA option and verify the effectiveness of the ICA by answering the questions: “Does it detect the issue?” and “Does it cause another issue?” with supporting data. Implement the ICA and validate its effectiveness in preventing the issue from reaching the customer using the same indicator(s) that made it apparent.
As long as the ICA is in place, monitor the effectiveness and take remedial action if nonconformities continue to pass the control point(s) or if nonconformities are found in reworked material.
Key D3 Elements:
- Defined ICA
D4: Diagnose Root Cause
Review the Is/Is Not Analysis created during D2, and identify differences between the Is and the Is-Not, but they could have been.
Review or create a timeline of changes made to the process, product, procedures, etc., and compare the timing of these changes to the timing of the issue.
Develop theories of possible root causes that could explain the differences and the timing. A tool that may be useful in developing theories is the cause-and-effect (fishbone) diagram.
Test the theories against the problem description. Look for theories that help explain the differences between the Is and Is-Not for the questions What?, Where?, When?, and How Big? If no single theory can explain the entire problem description, consider the possibility of multiple causes. A fault tree analysis is a helpful tool for developing multiple root causes.
Document the theories and the process of elimination using a diagnostic tree. Continue developing the diagnostic tree to approximately 4 - 6 levels deep. Stop when the level of detail becomes less actionable (i.e., outside local control).
When the diagnostic tree has been completed, prune the branches of all theories that have been disproven to create a 5 Whys (Problem causal).
When the 5 Whys (Problem causal) is complete, develop a 5 Whys (Escape causal) for the root cause of how the issue escaped local control points into the field.
Key D4 Elements:
- 5 Why (Problem causal)
- 5 Why (Escape causal)
- Diagnostic Tree
D5: Identify Solutions (Planned PCA)
Establish decision criteria for permanent corrective action(s) (PCA) to address both the root cause of the problem and the escape root cause. The decision criteria will consist of Must Have (i.e., mandatory, measurable, and realistic) requirements and Nice to Have (i.e., desirable, flexible, and realistic) wants.
Identify possible corrective actions for both the root cause of the problem and the escape root cause. Compare the choices against the must-have requirements and discard the options that fail to meet the must-have requirements. Using a relative scale, evaluate the remaining choices against the Nice to Have wants.
Select the option that meets all Must Have requirements and best meets the Nice to have wants without creating an unintended consequence.
Verify through testing that the action will resolve the problem at the root cause level, allowing for variation in the frequency or patterns created by the cause. Evaluate the action over the full range of production variation and operating conditions.
Key D5 Elements:
- Permanent corrective action
- Verification of PCA
D6: Implement, Verify & Validate Corrective Actions (Actual PCA)
Develop an action plan for implementing the Permanent Corrective Action. The plan should include critical steps, identification and prevention of potential barriers, and contingency plans (including triggers and responsibilities for implementation).
Implement the Permanent Corrective Action plan, then remove the Interim Containment Action put in place during D3.
Validate the effectiveness of the PCA for both the problem and the escape using the same metric that previously identified the problem. Valuable tools for validation include SPC, capability studies, audits, etc.
Key D6 Elements:
- Implementation plan
- Contingency plan
- Validation of effectiveness
D7: Prevention
Review everything learned in steps D0 through D6, analyzing how the problem occurred and escaped.
Identify policies, procedures, methods, and systems that allowed the problem to occur and to escape. Identify temporary corrective actions that should have been replaced with permanent solutions.
Look for undocumented or non-standard practices, lack of coverage, or blanket policies with no exceptions.
Identify similar products that run through the same operation or process as the root cause variable, similar processes susceptible to the same root cause problem, and similar control points susceptible to the exact escape root cause.
Identify other potential root causes that could result in the same failure mode.
Use Fault Tree Analysis to identify other potential causes, followed by a Process FMEA to assess and manage the risk.
Identify and choose preventive actions that may be applied to other products, processes, or facilities, including improved controls, using the same “Must Have” and “Want” processes described in D5.
Document these preventive actions through revised PFMEAs, control plans, policies, procedures, instructions, and lessons learned.
Key D7 Elements:
- Implementation plan
- Updated PFMEAs, control plans, policies, procedures, and instructions
D8: Congratulate Your Team
Update the lessons learned database and recognize the entire team’s collective effort.
Verify The Root Cause(s)
The root cause must explain all known data. Any theories that pass the trial run are the most likely causes. If only one theory passes the trial run, then verify this theory as the root cause. However, more than one theory may pass the trial run. In those cases (and when practical and feasible), collect and analyze any missing data for uncertain theories and re-examine information to resolve uncertainties.
To test the theory, do the following:
- Ask, ‘Does this theory explain the symptoms and data? If so, how?’
- Test the theory against each condition
If a theory explains the problem but lacks the information necessary to explain why it happened, gather more data:
- Gather more data to prove or disprove these theories
- Test simple (single change) theories first
- Test highly complex or interactive theories last
If additional information reveals that a theory cannot fully explain why the problem happened, eliminate it from consideration. If gathering and evaluating additional information is not feasible, try to verify each remaining theory. Start verification with the theory that best explains the symptoms.
Once you have determined the most likely cause(s), verify that they cause the problem. Verification is the proof that you must confirm that you have identified the root cause. Verification is done passively and actively. Passive verification is done by observation:
- Look for the presence of the root cause without changing anything
- If you cannot prove the root cause, the identified cause is not the root cause
Active verification is done by manipulating the root cause variable:
- Implement and remove the root cause variable to make the problem ‘come and go’
- Both ‘coming’ and ‘going’ are essential tests to confirm the root cause
- There can be more than one verified root cause
Verify the root cause(s) by conducting with/without studies. Can you generate or recreate the problem?
The root cause is an actionable factor preventing future occurrences if corrected or controlled. The root cause is the original event(s), action(s), and condition(s) generating (directly or in cascade) an actual or potential undesirable condition, situation, nonconformity, or failure.
Determine Escape Point & Implement Controls
After you have determined and verified the root cause, you need to determine the location of the problem's escape point. An escape point is the point closest to the root cause, where the problem could have been detected but was not.
A control system is a method deployed to monitor the product or process to ensure compliance with requirements. It consists of responsibilities, procedures, and resources.
A control point is a location within the control system where the product or process is checked for compliance with the standards, e.g., in-process inspection, sampling, first article inspection, final inspection, etc.
A product or process may have multiple control points within the system. When you identify the escape point, you can work to improve or establish another method of inspection into the system to ensure that if problems occur, they will not go undetected. To understand how the problem escaped and to identify the escape point:
- Review the process; focus on the part of the process where the root cause occurred
- Determine if a control system exists to detect the problem
Developing a new control system must be considered part of the problem’s solution if none exists. If a control system currently exists:
- Identify the control point closest to the root cause
- Determine if the control point is capable of detecting the problem
If the control system is incapable, developing an improved system must be part of the problem solution. If the control point can detect the problem, then the control point is the verified escape point. Choose and verify permanent corrective actions for the root cause and the escape point.
Select the best permanent corrective action to remove the root cause and eliminate the escape point. Verify that both decisions will be successful when implemented without causing undesirable effects. Steps for permanent corrective actions (PCA) selection include:
- Establish decision criteria, e.g., what is feasible
- Identify possible actions;
- Choose the most appropriate permanent corrective action (PCA)
- Test and verify the permanent corrective action
- Re-evaluate the ICA & PCA for the escape point
Step 5: Implement Corrective Action
Implement Permanent Corrective Action
At this stage, the root cause of the problem should be clearly identified and validated. Set up an action plan with clear responsibilities and target dates. List the corrective actions and select according to time to resolve and the implementation efficiency.
Permanent corrective actions must address the root causes of occurrence and non-detection. In the case of identifying non-manageable root causes, it is helpful to develop risk-mitigating actions (e.g., an early warning system or monitoring of pre-indicators).
Implement all selected corrective actions when all root and contributing causes have been identified and their effects understood. Please verify that the planned actions were taken as scheduled and assess their effectiveness in permanently preventing undesirable conditions, situations, nonconformity, or failures from recurring.
Steps for permanent corrective action (PCA) implementation:
- Implement the permanent corrective action (PCA)
- Implement controls
- Evaluate the permanent corrective action (PCA) for the escape point
- Remove the immediate containment action (ICA)
- Perform validation
- Confirm with the customer that the symptom has been eliminated
To ensure the most effective corrective actions to address the most likely or critical root causes, operational and business constraints such as costs, lead time, difficulty of implementation, and resources are considered. Select solutions that optimize value and effectiveness for all stakeholders!
Implement the solutions that have been selected, and verify that all actions have been completed to schedule and that they have prevented the undesirable condition, situation, nonconformity, or failure from recurring. Plan and implement selected permanent corrective actions. Remove the interim containment action and monitor the long-term results.
Communicate the corrective actions and the action plan to relevant stakeholders (including internal and external customers). Obtain necessary approvals (e.g., for tests on customer property). Execute the action plan, observe the effects, and take the supporting actions as required.
Update Documentation
Change the management and control systems, such as work instructions or processes (as applicable), to prevent the same or similar problems from occurring again. The following documents should be considered for revision:
- Work instructions and procedures
- Process descriptions
- Training and instruction plans and documents
- Quality control plans, instructions, and records
- Design and process guidelines
Prevent Recurrence
Modify the necessary systems, policies, practices, and procedures to prevent the recurrence of this problem and similar ones. Make recommendations for systemic improvements as necessary:
- Review the history of the problem
- Analyze how the problem occurred and escaped
- Identify affected parties
- Identify opportunities for similar problems to occur and escape
- Identify practices and procedures that allowed the problem to occur
- Identify practices/procedures that allowed the problem to escape to the customer
- Analyze how similar problems could be addressed
- Identify and choose appropriate preventive actions
- Verify preventive action and its effectiveness
- Develop an action plan
- Implement preventive actions
- Present systemic preventive recommendations to the process owner
Serious consequences may occur when the underlying symptoms are not addressed, when the quick fix is accepted as a final, permanent solution. Excessive reliance on containment or emergency response action will create a repeating cycle.

Step 6: Monitor Corrective Action
Review Process
Establish a review process to ensure corrective actions are completed according to plan and remain effective over time by confirming that you have done what you have planned. Try adjusting the type and number or frequency of additional checks and audits to ensure the correction actions remain effective.
Monitor the implementation status of corrective actions and their effectiveness, including internal and external stakeholders. Appropriate tests may check the effectiveness of corrective actions. It might be helpful to define at least one specific KPI per action to measure the effect of each action.
Depending on the criticality of the problem-solving, governance needs to be defined with regular reviews: e.g., daily quick meetings can be held to update each other on the progress, including Top management when needed.
When the same problem has been identified or is suspected to occur on the same or similar products, processes, or data, the same corrective actions must be implemented and their effectiveness verified for all these additional products, processes, or data.
The owner of each corrective action, the team leader, and all team members should verify the effectiveness of the actions taken to date and, when relevant, the customer. Examples of verification methods include:
- Additional process monitoring until it is demonstrated that the process is stable and capable of consistently meeting requirements (recording and analysis of process parameters and/or product characteristics, SPC, etc.)
- Additional internal audits to precisely verify the effectiveness of the corrective actions
- Associated metrics show significant improvement resulting from the corrective actions
Examples of supporting evidence might include updated procedures, work instructions, control plans, etc., to show that any changes were defined. Evidence of effective implementation of the changes is also required, such as SPC data, inspection, training, audit, etc.
Ineffective Corrective Actions
If the root cause’s corrective action is ineffective, return to Step 1.5. Identify the root cause(s) and revisit the analysis process to check if the failure was in the root cause identification and the development of the solutions.
If they are effective, evaluate which containment actions may be eliminated (e.g., stop over the inspection and production, return to normal transportation means, etc.) without adversely affecting the product and process output. Record evidence of actions completed and associated results (what works and what does not).
Knowledge & Learning
To document analysis results and changes to make the corrective action permanent, capture and share learning with all the stakeholders to prevent similar undesirable conditions, situations, nonconformities, or failures from occurring on other products, production lines, factories, or suppliers.
Identify all that can be shared from experience and transferred across business units, production lines, factories, or suppliers to ensure that similar problems will not be repeated in new developments or design revisions.
Ensure that you get agreement from appropriate levels of management and other process owners and functions (internally and externally) to launch actions and verify they are implemented and effective.
Once the problem is characterized correctly, contained, analyzed, and corrected, the organization should gather key information inside a lesson-learned record for knowledge sharing within the organization.
Keep a lessons-learned register, which includes a summary of content and results of analyses, flow charts, databases, performance data, main actions and decisions, the location where detailed data can be retrieved, difficulties encountered when managing the issue, etc. Your organization should consider the following information for the lessons learned record:
- Identification of the Lesson Learned (e.g., reference and/or link with the problem identification number)
- Description of the problem using a 5W approach
- Description of the cause of the problem
- Description of the learning from this problem (e.g., the output of a 5 Why analysis)
- Identification of the organization’s functions and locations potentially affected by the learning
- Keywords to ease the search of Lessons Learned record covering specific topics (e.g., problem typology, product family, key processes)
The organization should share and use these lessons learned:
- While designing a product or launching a new project
- During the new people induction for knowledge acquisition
- During Process Review, make the process more robust and avoid having the same (or similar) problem, e.g., in another process/ product subject to identical or similar preconditions for an occurrence of this (type of) problem
Considering the recent problem and similar problems that could potentially occur can lead to better and more comprehensive prevention of the recurrence of problems.
Implementing Actions Elsewhere
When the decision is made to implement actions in other business areas, such as production lines, factories, or suppliers, which are not under the direct control of the response team, implementation and the verification of effectiveness are not necessarily the team's responsibility.
Escalation to Top management or transfer to another function (procurement, engineering, etc.) may be required to ensure proper leverage and action follow-up.
Management Review
Undertaken at least annually, an analysis of patterns of issues, problems, concerns, complaints, etc., is performed to determine if the event is isolated or repetitive and to present findings to top management.
Top management should review the analysis findings, assess the effectiveness of corrective and preventive actions, and determine the need for procedural refinements or other measures to ensure continual improvements.
Providing the number of improvement actions or the number of improvement actions that are opened or closed to the management review process is not sufficient in assessing the effectiveness of the processes.
Any observations, conclusions, and recommendations for further necessary action from the review must be recorded. If any corrective action must be taken, Top management should follow up to ensure that the action was effectively implemented.
This review would include assessing any opportunities for improvement of the manufacturing process, the management system, or the organization itself.
What Is A Corrective Action Report?
A corrective action report (CAR) is a formal reporting method that is used to capture information about repeated nonconformities or problems within a process, product, or system that can arise from:
- Internal issues and internal audits. During routine internal audits and inspections, processes, procedures, and work instructions may be identified as nonconforming. These can initially be documented in the internal audit report
- Processes that produce negative results and repeated defective outputs. Any process that does not produce an acceptable product or service should be reported by any employee through the initiation of the corrective action report
The quality manager usually reviews the issues raised by each corrective action report to identify the level of action required by entering the details into the corrective action tracker. Repeated nonconformities of the same nature or significant deviations from procedures or policies must be reported to top management for action and resolution.
Including a description of requirements (reference drawing, specification, standard as appropriate) is essential. A detailed description of the nonconformance should use sketches, drawings, specifications, etc., to help the understanding of the nonconformity.
Describe the issue using plain language, and list any relevant documents and names of others who may be aware. Use terms that are understood by all. It is vital to have a correct problem description to identify causes.
A corrective action report should be issued for repetitive, ongoing problems during our production processes and issues that impact customer end product delivery and quality management system noncompliance. Other nonconformities outside these criteria should also be controlled through the remedial action system.
All corrective action reports raised can be categorized as follows:
| Risk Level | Recommended Problem-solving Method |
| OFI | Improvement opportunities that do not need correction can be enhanced, improved, or made more efficient. Use the F1030-01 Improvement Form. Complete within 10 Days. |
| Low | A low-level problem typically closed to immediate correction requires containment and trending. Use the F1020-02 A3 Action Plan. Complete within 15 Days. |
| Medium | A problem that results in a moderate impact. Requires containment and root-cause analysis, as well as corrective actions when appropriate. Use the F1020-03 5-Whys Worksheet. Complete within 30 Days. |
| High | A problem that results in a major/significant impact or is repetitive. It requires a root-cause analysis and corrective and preventative action(s). Use the F1020-04 8D Worksheet. Complete within 60 Days. |
Where applicable, any corrective action taken and controls implemented to eliminate the cause of nonconformity are applied to other similar processes. They are controlled by the quality manager, who liaises with affected process owners. Significant actions must be entered into the corrective action tracker.
What Is A Corrective Action Tracker?
The corrective action tracker is a spreadsheet demonstrating a commitment to continuous improvement and proactive problem-solving. It systematically manages and resolves organizational issues, defects, or nonconformities. It helps ensure accountability, track progress, and prevent problems from recurring, while aligning with the ISO 9001 corrective action requirements.
Our corrective action tracker tool lets you quickly determine each nonconformity's course of action and monitor its status in real-time. This ensures the appropriate tools and techniques are applied for problem-solving and formulating corrective actions.
Based on the information captured from the corrective action report, the corrective action tracker prompts the user to use 1 of 3 increasingly detailed problem-solving methods (e.g., A3, 5-whys, or 8D), depending on the severity or complexity of the nonconformity.
The tracker will determine which problem-solving method is appropriate to close-out the nonconformance. Use the suggested problem-solving forms to diagnose the nonconformance and update the corrective action plan with the relevant details.
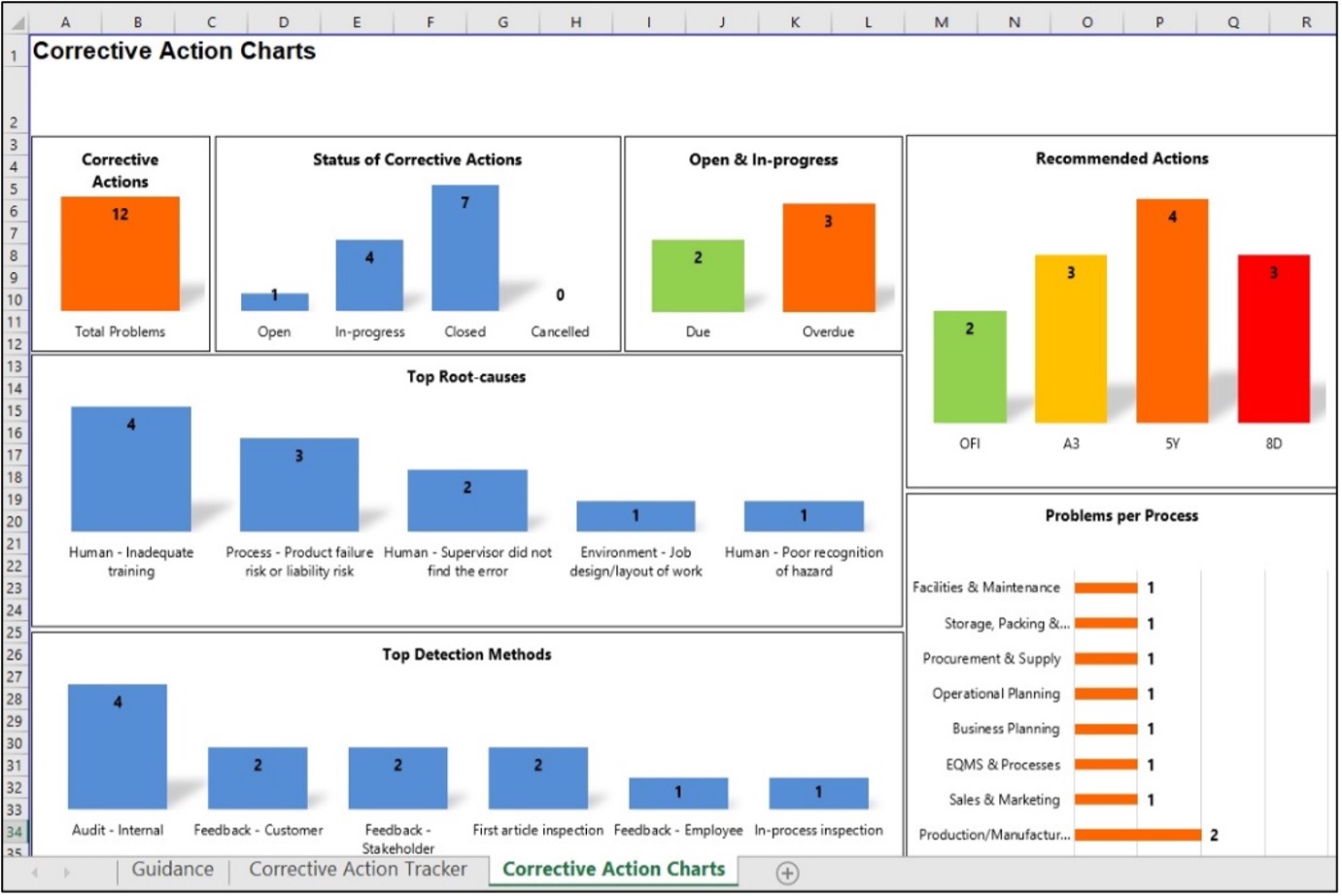
The corrective action charts deliver detailed reports and summaries, keeping all parties informed and engaged. The charts in the 'Corrective Action Charts' tab will automatically update based on the database in the 'Corrective Action Tracker' tab. To locate the data for each chart, right-click the chart and click 'Select Data' from the menu.
This will open the tracker tab and highlight the source data, allowing you to adjust the data range each chart displays manually.
Using The Corrective Action Tracker
Problem Definition
Use Part A to enter data from Section 1 of the Corrective Action Report in the fields below in Columns B to K. How serious is the problem? Enter the perceived risk level in Column H, AND choose ONE option from Columns I, J, or K.
- Enter the Data from the Corrective Action Report
- Capture the data from the corrective action report in Part A to determine the most suitable problem-solving method to address the causes of the nonconformance
- From the corrective action report, enter the details from Section 1 into the related fields in the tracker
- Based upon judgment and experience, categorize the perceived risk level of the problem using the drop-down menu in Column H
- Classify the problem by choosing ONE option from either Column I, Column J, or Column K
- Entering Y’s or N’s results in a risk score that ranges from 1 to 4
- Column M will calculate an overall risk score based on the data previously entered
- The suggested problem-solving method shown in Column N is based on the following trigger scores: 1 = OFI, 2 = A3 (Who, What, When action plan), 3 = 5Y (5-Whys cause analysis), and 4 = 8-Disciplines (In-depth analysis)
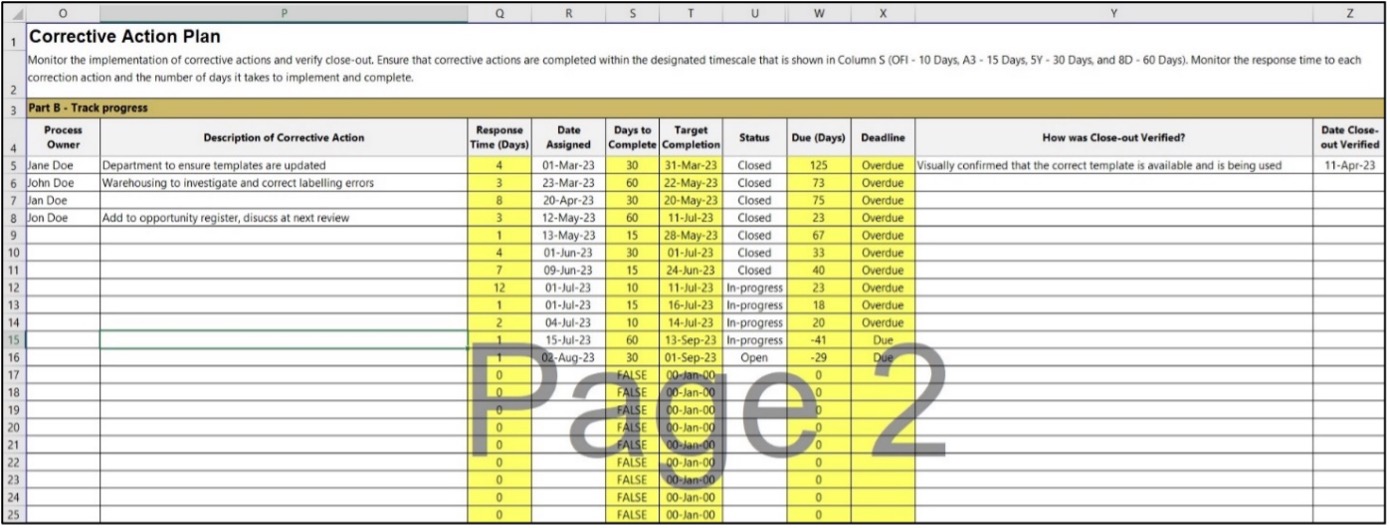
Corrective Action Plan
Monitor the implementation of corrective actions and verify close-out. Ensure corrective actions are completed within the designated timescale shown in Column S (OFI - 10 Days, A3 - 15 Days, 5Y - 30 Days, and 8D - 60 Days).
Monitor the response time to each correction action and the days it takes to implement and complete.
- Undertake root cause analysis
- Issue the chosen root cause analysis method to the process owner/response team for completion
- Once completed, update the root cause in Part A
- Update the 'Corrective Action Plan' in Part B
- Monitor the implementation of corrective actions and verify close-out
- The status of the corrective action will remain 'Open' until work to correct the nonconformance begins, then the status becomes 'In-Progress'
- This means the status stays 'In-Progress' until the associated corrective action is verified. The status of the nonconformance would then change to 'Closed'
- Should the corrective action request be withdrawn, the status is set to 'Cancelled'
Input Messages
Amend or delete the input message, highlight the relevant cells, select the 'Data' tab, select and click 'Data Validation' from the drop-down menu, then select the 'Input Message' tab. Amend the 'Input Message' in the dialogue box, or select 'Clear All' to remove the input message.
Drop-down Menus
Use the drop-down menus in Column B to select and enter a Process Name from the list. You should modify the process names to match those used in your organization by amending the source list in Column AB.
Use the drop-down menus in Column E to enter the detection method. You can modify the detection list by amending the source list in Column AD.
Use the drop-down menus in Column G to enter the root cause. You can modify the root cause list by amending the source list in Column AF.
Status of Corrective Actions
The corrective action status will remain ‘Open’ until work to correct the nonconformance begins, then the status becomes ‘In-Progress’.
This means the status stays ‘In-Progress’ until the associated corrective action is verified. The status of the nonconformance would then change to ‘Closed’.
Should the corrective action request be withdrawn, the status is set to ‘Cancelled’.
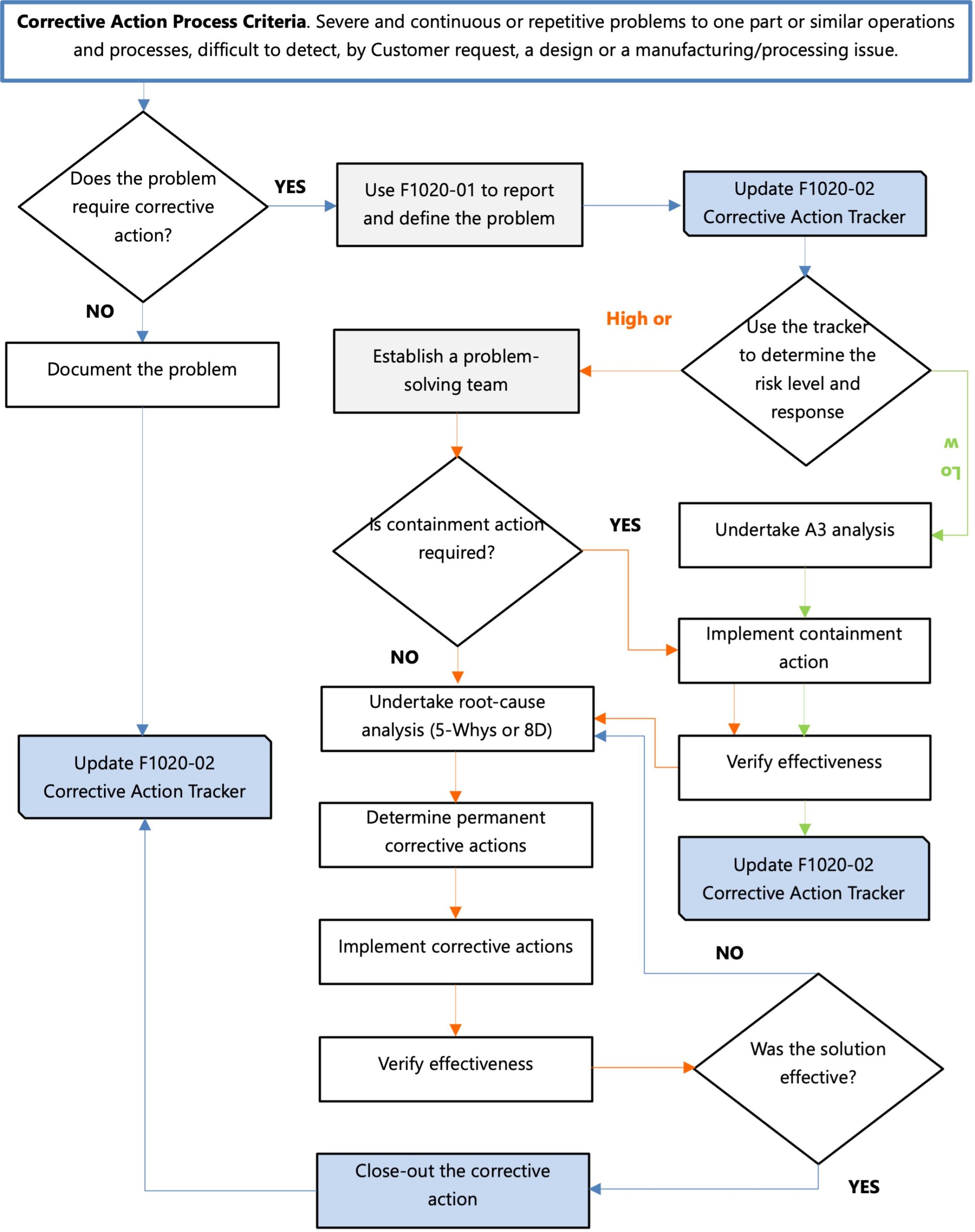
What Is A Corrective Action Process Map?
Using a top-down approach, the corrective action process map describes how the process steps are structured into a hierarchy of activities that show the sequence and the responsibilities for each step or task.
Summary
| Corrective Action Steps Summary | |
|---|---|
| Define the Problem | Identify the internal/external customer problem in quantifiable terms; define, verify, and implement interim containment action to isolate the problem's effects. |
| Establish an Investigation Team | Form a team with process/product knowledge, allocated time, authority to solve the problem, technical skills, and a designated team leader. |
| Select an Interim Containment Action | Choose an interim containment action, kept in place until a permanent action is verified; this action must protect the customer without introducing new problems. |
| Verify the Interim Containment Action | Ensure the interim containment action effectively protects the customer from the problem and does not introduce new issues. |
| Identify the Root-Cause | Isolate and verify the root-cause by testing each possible cause against the problem description and data. |
| Complete a Comparative Analysis | Describe the problem in terms of what, where, when, and how big; consider features such as people, processes, materials, machines, and the environment; list all facts and changes to determine root-cause. |
| Develop Root-cause Theories | Generate theories on how the problem occurred using brainstorming; consider each change and list theories for each, without dismissing any possibility. |
| Test the Theories | Test each theory against the symptoms and data, gather more data if necessary, starting with simple theories and moving to complex ones. |
| Verify the Root-Cause | Confirm the identified cause(s) actually cause the problem through passive observation and active manipulation of variables; both ‘coming’ and ‘going’ of the problem must be confirmed. |
| Determine and Verify the Escape Point | Identify the escape point where the problem could have been detected but was not; review the process and determine if a control system exists or needs to be developed to detect the problem. |
| Implement & Validate Permanent Actions | Plan and deploy selected permanent corrective actions; remove interim action and monitor long-term results; evaluate the action for the escape point and perform validation. |
| Prevent Recurrence | Modify systems, policies, and procedures to prevent recurrence of the problem; identify and implement preventive actions, verify their effectiveness, and make systemic improvement recommendations. |
Related Information You Might Find Useful
- 10.2 Nonconformity in ISO 9001
- 10.2 What is Non-conformance?
- 10.2 Nonconformity and Corrective Action [ISO 45001]